
Automated Breast Ultrasound (ABUS): Clinical Utility, Evidence & Integration
An evidence-based overview of ABUS as a supplemental imaging modality in women with dense breast tissue and its application in clinical referral pathways.
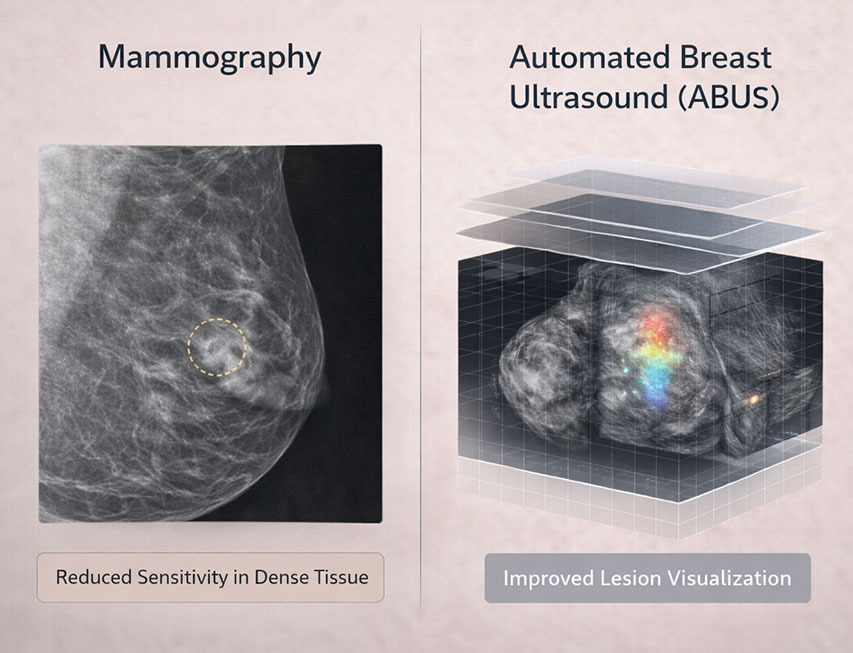
Note: ABUS is intended to supplement mammography when indicated and is not recommended as a stand-alone screening tool in routine practice absent specific clinical justification.


ABUS is the only FDA-approved supplemental ultrasound screening technology for women with dense breasts.

ABUS adds diagnostic sensitivity but may increase recalls and biopsies; clinical judgment is essential.

Evidence supports enhanced cancer detection when used in conjunction with mammography in appropriate populations.

Integration into practice requires breast density reporting and appropriate case selection.